A treatment gap closes for the youngest patients
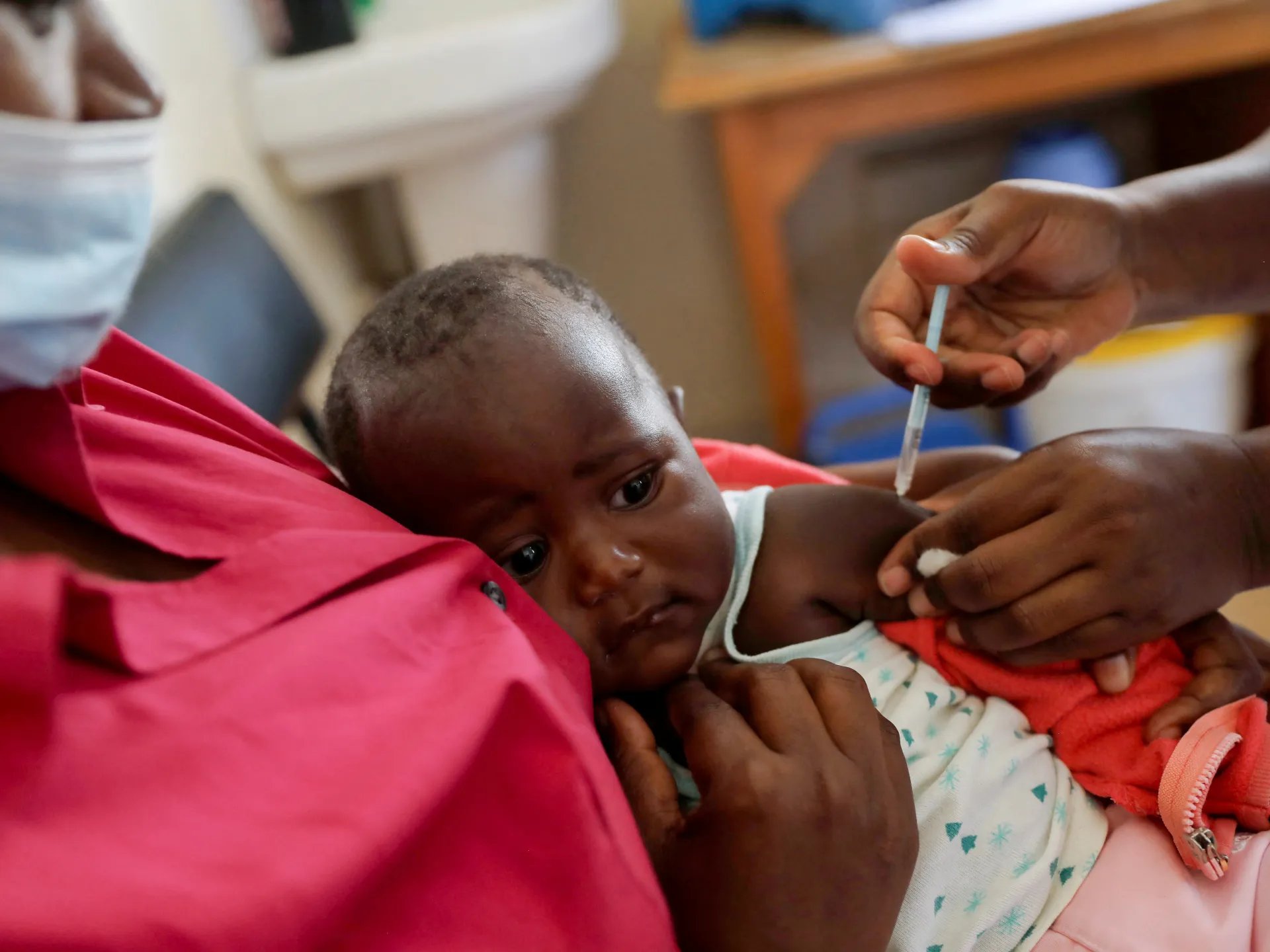
The World Health Organization approved artemether-lumefantrine on Friday, marking the first antimalarial drug formulated specifically for newborns and infants. The approval addresses a critical gap in pediatric malaria care: until now, babies have received medications designed for older children, exposing them to risks of dosing errors, side effects, and toxicity.
The drug is safe for infants weighing less than 5 kilograms (2.2 pounds). WHO chief Tedros Adhanom Ghebreyesus said the approval signals a shift in how the disease is being tackled. "For centuries, malaria has stolen children from their parents, and health, wealth and hope from communities. But today, the story is changing. New vaccines, diagnostic tests, next-generation mosquito nets and effective medicines, including those adapted for the youngest, are helping to turn the tide."
Why infants face the highest risk
Children under five account for around 70 percent of all malaria deaths globally. The disease also devastates pregnant women and newborns: malaria contributes to an estimated 10,000 maternal deaths annually, 200,000 stillbirths, and around 550,000 babies born with low birth weight, according to the WHO.
Sub-Saharan Africa accounts for nine out of ten malaria cases and deaths globally.
What the approval enables
The prequalification designation means the medicine meets international standards of quality, safety, and efficacy. The approval allows countries to authorize the drug even without the capacity to conduct full clinical trials. UN agencies can now procure and distribute the treatment in malaria-endemic areas, subject to government approval.
This streamlined process accelerates access in regions where clinical infrastructure is limited but disease burden is highest.
The broader malaria fight
Malaria remains a major global health challenge, with about 282 million cases and over half a million deaths reported in 2024. Yet progress has been documented: since 2000, global efforts have averted an estimated 14 million deaths.
Earlier this April, the WHO approved three new rapid diagnostic tests after malaria parasites evolved to become harder to detect. In parts of the Horn of Africa, up to 80 percent of cases were missed due to false negatives, delaying treatment and increasing death risk. In 2021, the WHO recommended the first malaria vaccine, which has since been introduced on a large scale across multiple African countries.




































































































































































































































































































































































































































The sources also report that the WHO's approval allows UN agencies to procure and distribute the treatment in malaria-endemic areas, subject to government approval.